General hygiene
Good hygiene is paramount to good results. Not everyone has modern, state-of-the-art designer hatcheries but with attention to detail the results can be as good. Starting with personnel, they should be provided with shower facilities and clean clothing daily and showering must include the head. The clean clothing should be placed beyond the shower and the whole ablution block should always be kept very clean. Staff must not leave the building until they stop work.
Visitors should be minimal and must also go through the same process. This must include persons brought in to mend and maintain equipment if it is beyond the experience of your own staff.
In the hatchery itself each section should be kept separate with mats soaked in disinfectant in each doorway and plastic doors will help to reduce problems and keep the airflow in different rooms separate. In many hatcheries now, rooms are separated by solid doors but unless controlled with sensors they are often left open which alters the pressure and compromises biosecurity. The more sophisticated the system and design the easier this is but on the other hand there is far more to go wrong and far more nooks and crannies to keep clean, air socks, conduits, pipes, tunnels etc.
The frequency of opening doors should be minimised to prevent air drawing from one room to the other. In critical areas of the hatchery, the air is maintained at a positive pressure, so contamination can not be drawn in through an open door. Doors, including one-way doors, help stop cross contamination between rooms.
Chick fluff can be a problem to human health and good airflow and a specially designed fluff tunnel will remove fluff which is also a carrier of any disease organisms present in the hatchers. Samples of fluff and also samples of meconium should be regularly monitored in your own or a veterinary laboratory.
The hatchery rooms must be kept clean and this includes all surfaces including the ceilings, ledges and tops of machines. Each room and each machine should be cleaned after use and contact plates should be examined regularly by yourselves or veterinary services to identify any problems. Many hatcheries do not have good enough facilities for filtering air so that bacteria and fungal spores can easily be drawn into the system. The latter can be a problem in some climates and vigilance is necessary. Good monitoring will tell you precisely where a problem might be developing and then you can take action.
It is as well to remember that staff can be carriers of many potential disease bacteria and viruses although it does not affect them in any way. Some hatcheries make a point of checking staff and if a problem is found that person can be kept out of the buildings until treatment has been effective.
As staff do not leave the building for meals it is usual to supply a canteen. In some hatcheries mixing of staff from one section to another is forbidden yet at mealtimes they all come together!
In a clean hatchery problems are unlikely but washing hands before and after using the canteen and strategically placed disinfectant mats in the doorways will minimise any disease problem. Large modern hatcheries are equipped with separate canteens for the staff working in the clean or the dirty area in order to avoid the crossing of people.
As with the ablution block, the canteen should be cleaned daily and waste etc. removed. The canteen should be after the shower block so that the staff can leave their food there before entering the main building.
Develop a good relationship with your veterinary services and be aware of regulations in your own country which might affect the hatchery.
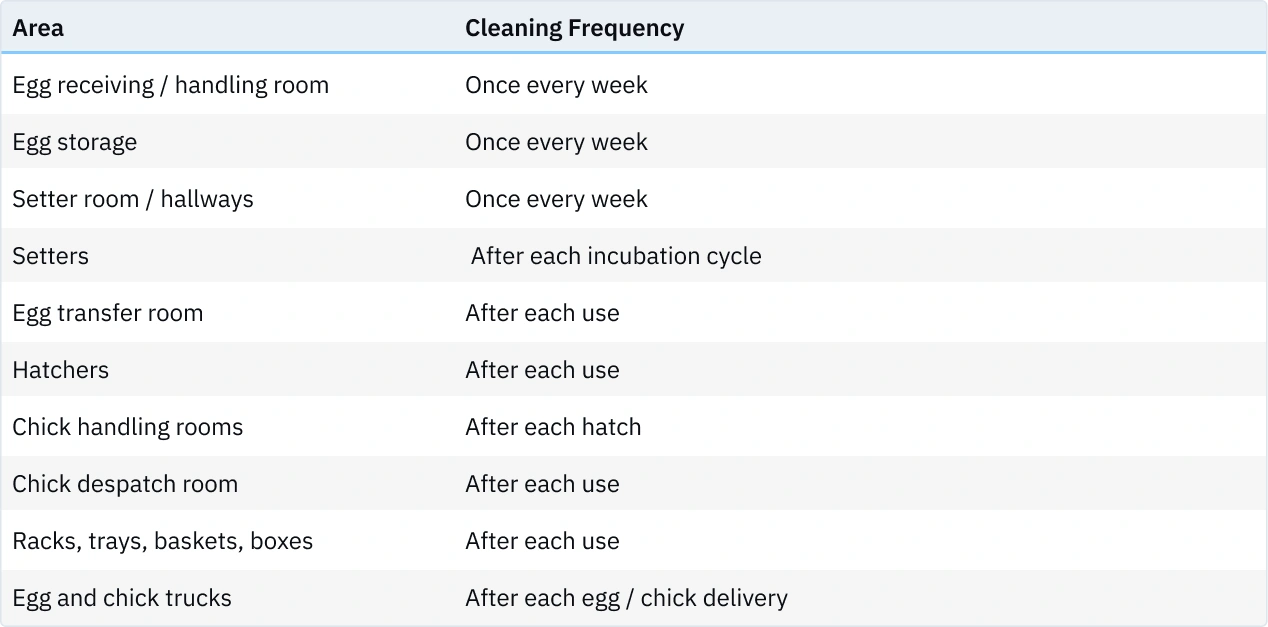
Cleaning
For every hatchery room, instructions for cleaning and disinfection should be formulated and pinned up in the particular room. When formulating instructions for cleaning rooms and equipment, keep the following aspects in mind:
- First, all debris such as fluff, blood, eggshells, broken eggs and dirt needs to be removed. Since organic material inhibits the chemical action of disinfectants, it is very important that all surfaces to be disinfected are free from debris before applying the disinfectant. Depending on the degree of soiling this is done by:
- First dry cleaning with a vacuum or sweeping.
- Soaking of the surface with a foamcleaner. PH of the detergent should be changed periodically (for example 3 weeks alkaline, 1 week acid) to remove potential biofilms and mineral deposits
- Rinsing with water
- Allow to dry.
- After the surfaces are clean and dry apply disinfectant. A wet surface will dilute the disinfectant and reduce its efficacy. the label closely and follow instructions. Factors affecting the efficiency of disinfectants are: contact time, temperature, concentration, pH, nature of soiling and compatibility with detergents.
- First, all debris such as fluff, blood, eggshells, broken eggs and dirt needs to be removed. Since organic material inhibits the chemical action of disinfectants, it is very important that all surfaces to be disinfected are free from debris before applying the disinfectant. Depending on the degree of soiling this is done by:
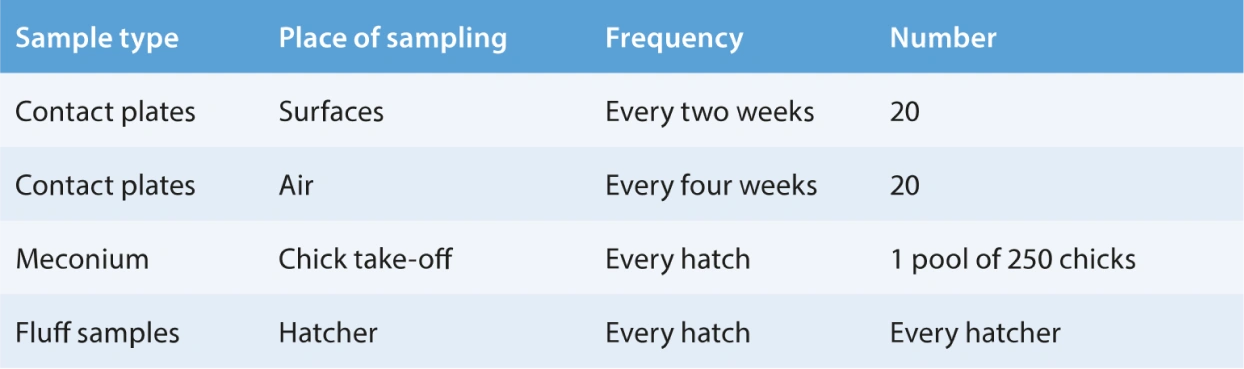
Hatchery microbiological monitoring
Incubation Guide Layer Version 2008, Pas Reform.
Microbiological monitoring is an essential element of any hatchery quality assurance programme for the evaluation of cleaning and disinfection procedures. It is crucial to conduct the monitoring programme regularly. Here, guidelines are given describing basic monitoring procedures to evaluate the success of hatchery cleaning. A more intense monitoring programme should be used to solve a specific problem and would include egg washes, chick sampling and bacterial identification.
To evaluate the effectiveness of a sanitation programme, the assessment of cleanliness and sanitation should be performed only after clean up has occurred. For microbial monitoring, use is often made of solidified agar in Petri dishes containing nutrients that match the metabolic needs of bacteria and fungi. Bacteria and fungi grow on this media when put in an incubation cabinet. They will multiply and become visible as colonies. The number of these colonies indicates the hygienic state of the surface or air sampled.
Flat surfaces
Rodac plates are plastic plates of which the bottom side is filled with agar gel. This agar layer is slightly higher than the edge of the plate so that direct contact is made with the surface to be sampled. Remove the cover of the plate, press the agar gently upon the surface to be monitored (do not move while contact is made). The cover should be replaced after the impression is made, taking care not to touch the agar.
To detect fungi one can use Sabarrouhd plates in the identical manner.
Air
The same plates can be used to monitor the microbiological status of the air. Expose the plate with the selected media, by carefully placing the plate, media half on the bottom, on a flat surface within the environment to be monitored. Gently remove the cover and let the plate rest. For relatively clean areas, a 10 minute sampling time is sufficient.
The agar plates which are being evaluated for bacterial contamination should be incubated for 48 hours at 37–37.5 °C in a microbiological incubator or a setter (place the plates in a plastic bag and set where they will not be disturbed). Plates are incubated upside down so that drops of condensation will not fall on the inoculated surface.
After incubation, the colonies on the agar media can be counted and recorded. The evaluation and monitoring of the hygiene conditions should be based on the hatchery’s own criteria. In general, excessive colonies indicate poor sanitation procedures or a hatching egg production problem.
For detailed advice on sampling, reading and evaluating agar plates, see instructions and advice of manufacturers of agar media.
It is advisable to maintain records of all results so that changes occurring over time can be observed in the different areas monitored. Also, the results should be carefully compared with liveability data.
Do the basics right
- Make sure staff are trained and understand the importance of good hygiene.
- Check that incoming eggs are clean.
- Check that soap/hand disinfectant are available daily and that they are being used.
- Check that mats are soaked in disinfectant.
- Physically check for accumulated dust/ debris on surfaces at all levels.
- Physically check that all filters are cleaned regularly.
- Check that all doors are closed between rooms.
- Physically check equipment after cleaning.